Tag: bioequivalence
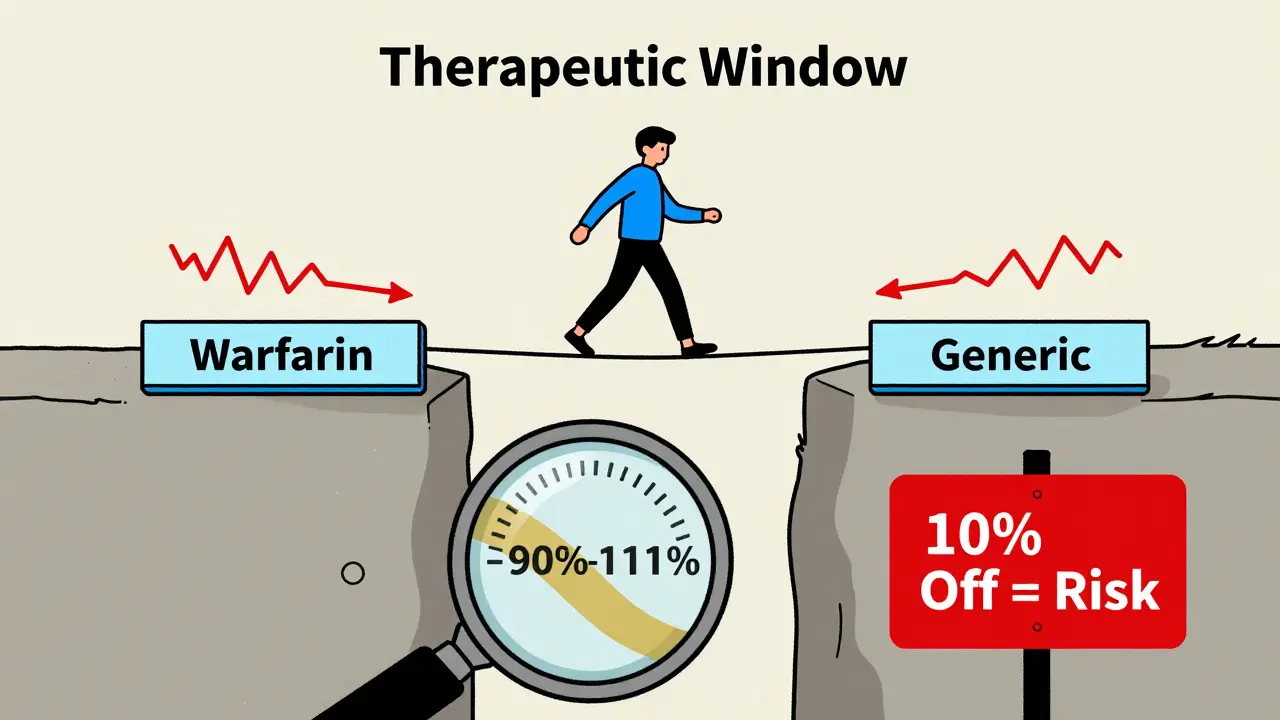
Bridging Studies for NTI Generics: Ensuring Safety and Efficacy
Bridging studies for NTI generics ensure that life-saving generic drugs like warfarin and levothyroxine are as safe and effective as brand versions. These complex, costly studies tighten bioequivalence standards to protect patients from dangerous dosing errors.
View More
Replicate Study Designs: Advanced Methods for Bioequivalence Assessment
Replicate study designs are essential for assessing bioequivalence of highly variable drugs. They reduce sample sizes, improve success rates, and enable regulatory acceptance through reference-scaling. Learn which design to use, how to avoid common pitfalls, and what the industry is doing in 2026.
View More




